PSYCH Symposium returned at the British Museum on 6 July to explore how psychedelic medicines can innovate healthcare – from mental health to eating disorders and beyond.
Welcoming hundreds of delegates PSYCH Symposium saw insightful discussions, one on one interviews and workshops exploring different aspects of the psychedelic industry – from the importance of trained therapists to the challenges faced in the drug development pipeline.
Shortwave Pharma’s CEO Rivki Stern kicked off a packed day of sessions with an address to the hundreds of industry stakeholders, medical professionals and investors in the audience.
“Every time that I visit the UK or Europe, I observe the duality, the coexistence of old and new… And I believe that this unique combination of tradition and innovation that is so characteristic of the UK and Europe also defines our industry today,” she began.
Stern explained that, on the one hand, the field of psychedelics is breaking ground in the mental health sector, which has seen little innovation for decades, however, on the other hand, none of this innovation could take place without adhering to the strict and traditional regimen of scientific exploration.
With government and even the pharmaceutical industry’s interest now piqued, she said the industry was now on the way to commercialisation.
Psychedelics for eating disorders

To put the real-world potential of psychedelic treatments into perspective, the first session focused on encouraging early research into the treatment of eating disorders, which have increased 140% over the last decade.
Stern, who was part of the panel, laid out the extent of the issue, suggesting that eating disorders currently cost the US economy around USD$65 billion a year.
Clinical Psychologist at Sheba Medical Center Yoel Golbert added that ‘the reality is dire, there are lots of patients calling out for quality care, but the reality is we don’t have much to offer’.
Oxford University’s Professor Rebecca Park explained that with conditions such as anorexia nervosa, neurally patients have a very rigid system, with a “desync” between the brain and body which makes it very easy for people to starve to death.
The panel then explained how both psilocybin and MDMA showed potential in being able to “break this desync” and help encourage much-needed neuroplasticity.
Golbert said: “There are encouraging indications that brain connectivity and activity create this window of neuroplasticity which can break the inherent rigidity of anorexia. It opens up a window that we can work with through therapy and through integration.
“MDMA also can help with anxiety-reducing properties. It’s an inducement of self-compassion and self-connection.”
Challenges in drug development
Next, the focus turned to the unique challenges presented by psychedelic drug development and treatment.
Cybin’s Chief Medical Officer, Dr Amir Inamdar, explained that while psychedelics were not unique in terms of drug development, the fact that they are not taken on a daily basis, they alter consciousness and they are usually taken alongside psychotherapy means ‘they present unique challenges’.
Because of these unique characteristics, he said that the gold standard, double-blind placebo studies are ‘virtually impossible’, and that his company had had to make ‘certain decisions to that design’.

Furthermore, Inamdar stated that he was “confident” big pharma and institutional investors would eventually get behind psychedelics “because the economics make sense”.
He argued that traditional SSRI and CBT treatment represented a “constant drain on resources”, whereas psychedelic treatments needed a small number of sessions that were more intensive.
“Having been in drug development for over 20 years, I’ve never been so excited. I’m as excited as a child right now.
“Almost all of us, in some form, have been touched by mental health issues. We’ve been giving out treatments for 50/60 years now, which don’t seem to do much. Here is an opportunity to change how we see and feel about mental health.”
Ketamine-assisted psychotherapy
The following session focused on the potential of ketamine-assisted psychotherapy, which Professor Celia Morgan suggested was often seen as the ‘dodgy uncle of psilocybin’.
Despite its reputation, Professor Morgan said ketamine has a number of unique properties and presents opportunities other compounds don’t.
“It has some useful properties, and one of those is that it’s probably the most common useful analgesic in the world. It’s something that is known to physicians and I think in that way, it is quite accessible – and we know that clinicians are risk averse,” she said.
In a presentation of her preliminary research, Morgan explained that early indications of ketamine’s ability to treat Alcohol Use Disorder (AUD) found that there was a significant increase in abstinence for those treated with ketamine.
In a separate study, she suggested that esketamine was found to significantly increase participants’ engagement and willingness to participate in mindfulness practice, while also helping to limit cravings for alcohol.
“The ketamine cat is already out of the bag, particularly in the US. It has the potential to be the groundwork for the introduction of psychedelics into mainstream medicine, but it could also be a drag on all the other psychedelics.”
Access to treatments
Following the recent significant developments in Australia, which are set to give mental health patients access to psilocybin and MDMA, the next panel focused on the recent progress and remaining barriers to accessing psychedelic treatments.
While the panel agreed that these compounds were overdue a reassessment of their current classification given their clear medical potential, they suggested that one of the biggest barriers was reimbursement.
Professor Chris Langmead, Deputy Director, of Neuromedicines Discovery Centre, told the audience: “At the moment, there is no reimbursement available through the Australian healthcare system.
“So, all of this will essentially have to be privately paid for, and that is not a sustainable situation if the global mission is trying to make these therapies as accessible as possible for the patients that really need them.
“We need to be leveraging the breakthroughs we are having now to really move the dial.”
Facilitating psychedelic investment
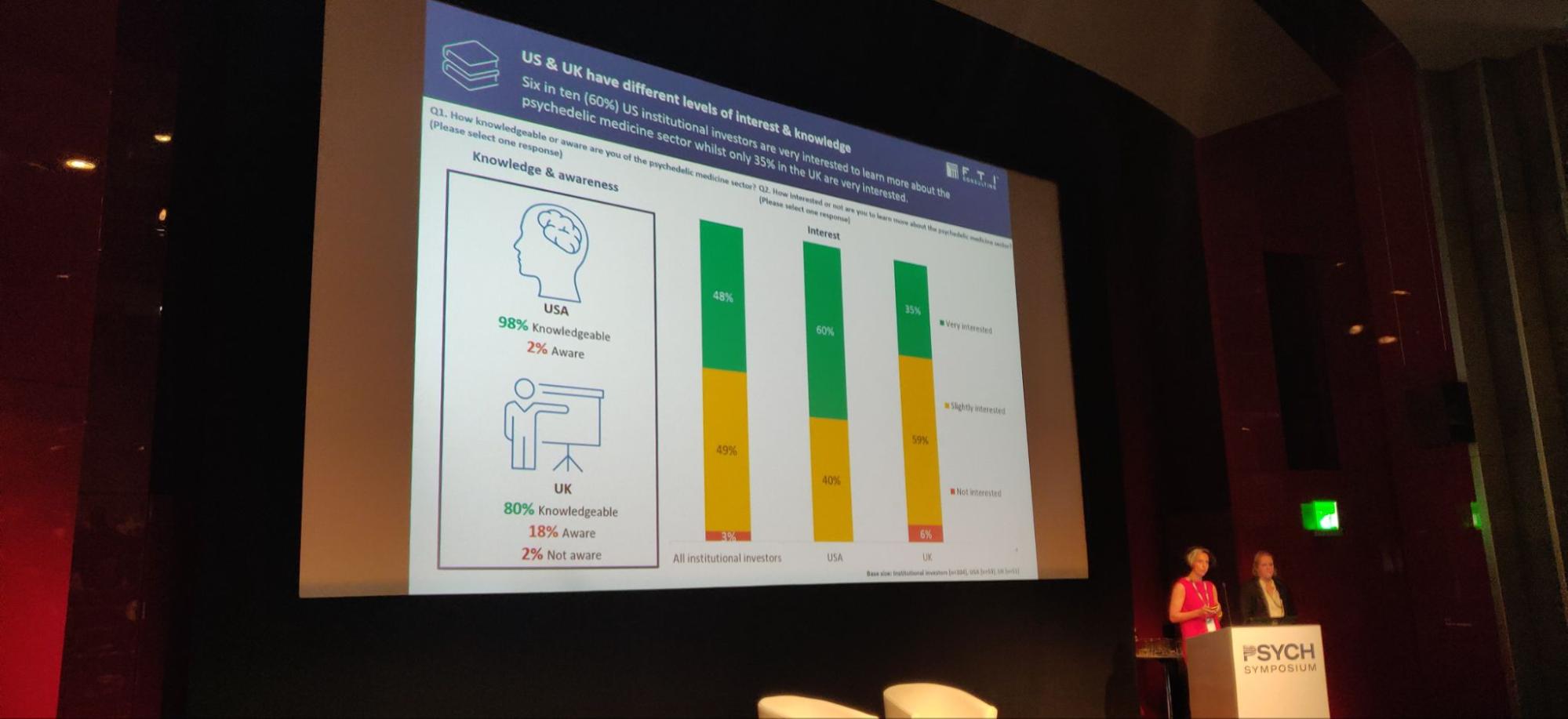
The following sessions of the morning turned to attracting investment into the psychedelics space, starting with a presentation of a recent survey conducted by PSYCH Symposium sponsor, FTI Consulting.
According to its survey of 104 international institutional investors, who managed a collective USD$10 trillion, there was ‘interest across the board’ in the psychedelics sector.
However, investment remained thin on the ground due to residual issues with public perception, poor company management and a lengthy return on investment.
This was echoed by Apeiron Investment Group’s founder Christian Angermayer, who said that, in a nutshell, institutional investors simply “don’t think the stock will go up”.
He continued that this was due in large part to the poor performance of the biotech sector during the past 18 months, which has been hit hard by growing inflation because of its often extended periods of loss-making before generating revenue.
Crucially, however, big pharma companies are “not sure what to make” of psychedelic treatments because they stray so far from the traditional model of selling “pills to patients every day”.
“We have to prove to ourselves that we can make this new paradigm work.”
Angermayer said he was more optimistic than many investors because he believes the rollout will be quick and organic.
“I have a very optimistic view because we have that infrastructure already there, because we have therapists. It’s practically just a bit of an add-on to traditional therapy, with a little bit more training.”
Additionally, he said that from personal experience everyone he knew who had tried psychedelics as a treatment found not only that it was effective, but also that it immediately turned them into an advocate.
“Why is this so important? Because once these medications are approved, we’re gonna see an unprecedented bottom-up drive. I think that is the number one issue which will determine the success of psychedelics – patients will demand it from their doctors.”
The future of psychedelic medicine in the EU

The final session of the morning saw some of Europe’s leading progressive politicians discuss the ‘The Future of Psychedelic Medicine in the EU’ in a session led by Psychedelic Health’s Stephanie Price.
Despite Albert Hoffman discovering LSD in Switzerland in 1938, the scheduling of psychedelic substances on the continent has seen 50 years of scientific censorship.
However, with numerous clinical trials now being carried out, policymakers in Europe are now starting to take notice.
Panelists agreed that the outlook in the EU was changing due to the growing prevalence of psychedelic clinical trials, and growing clinical evidence showing the efficacy of psychedelics as potential mental health treatments, however, they empahsised one of the key barriers to the rollout of these therpie in Europe was the ‘disparity of healthcare systems across the EU’.
Cyrus Engerer, MEP for Malta, explained: “In the EU, we have one single market when it comes to all other products. However, when it comes to pharmaceuticals and medicine, we have 27 separate markets, and that does not make sense.
“The fragmentation of the European market leads also to exclusivity. So, certain medicines are exclusive to certain patients only across the European Union, and that is unacceptable. It should be their right to access these treatments.”
The panel said that this framework is now on the verge of being overhauled with the introduction of a new European pharmaceutical revision – the first in 20 years – aiming to make access to medicines easier and more affordable, and to foster innovation in the medicines space.
Tadeusz Hawrot of the Psychedelic Access and Research European Alliance (PAREA) empahsised the significance of the review, noting that: “The ambition is to normalise access across the EU. And the second goal is stimulating innovation.”
MEP Mikulas Peksa – Czech biophysicist and member of the Pirate Party – emphasised that the main hurdles to the rollout of psychedelic medicines and therapies in the EU was the UN’s scheduling of substances such as psilocybin and LSD.
Whether this will change remains to be seen – but the UN this year, for the first time in its history, has included a section on psychedelics in its Word Drug Report 2023. This, coupled will the European Medicine Agency’s response to a cross-party call to be more active in psychedelics – leading to a psychedelics workshop being held in the European Parliament this year – may reinforce the changing attitudes towards these substances that have suffered decades of stigmatisation.
Reporting by Ben Stevens and Stephanie Price.

 Opinion2 years ago
Opinion2 years ago
 Insight3 years ago
Insight3 years ago
 Medicinal2 years ago
Medicinal2 years ago
 Research2 years ago
Research2 years ago
 Medicinal2 years ago
Medicinal2 years ago
 Markets & Industry1 year ago
Markets & Industry1 year ago
 News3 years ago
News3 years ago
 Medicinal2 years ago
Medicinal2 years ago




















