Psychedelics, also known as hallucinogens, are a class of psychoactive substances that alter perception, mood and cognitive processes. They work by interacting with specific receptors in the brain and altering the way that the brain processes information.
Classic psychedelics work by activating the serotonin receptors in the brain. Serotonin has a wide variety of functions in the human body including cognition, perception, emotions, appetite and digestion.
People sometimes call it the “happy” chemical because it contributes to well-being, mood and happiness.
The scientific name for serotonin is 5-hydroxytryptamine (5-HT). It is a neurotransmitter, meaning that the body uses serotonin to send messages between brain cells.
One of the primary ways that psychedelics work is by activating serotonin receptors in the brain, particularly the serotonin 2A receptor (5-HT2A). Activation of the 5-HT2A receptor by psychedelics can result in changes in activity of serotonin and other neurotransmitters, such as dopamine and norepinephrine, which can contribute to the drug’s effects.
The 5-HT2A receptor and the binding of classic psychedelics to it are well studied, which allows chemists to model the relationships between the activity caused by a psychedelic and its structure. Recent efforts have been made to use computational modeling to engineer psychedelic-based therapeutics to increase the diversity of psychedelic compounds and also to engineer compounds that lack hallucinogenic effects, but maintain the therapeutic benefit.
The specific effects of psychedelics can vary widely depending on the chemical structure of the psychedelic, in addition to the dose and the individual’s unique brain chemistry.
Level up your chemistry knowledge
All classic psychedelics have a similar chemical structure, we call this a chemical scaffold or a pharmacophore. A pharmacophore is like a key. Just as a key is specifically shaped to fit into a particular lock, a pharmacophore is specifically shaped to fit into a biological target, like the 5-HT2A receptor.
In the figure below, you can see that the basic pharmacophore is a 6-pointed ring with a small chemical “arm” on the right. The pharmacophore is basically a skeleton key that can bind and activate the 5-HT2A receptor, and then the additional chemical groups that make each psychedelic unique can dictate the potency of the drug, duration of the psychedelic experience and types of hallucinogenic effects.
Differences in this basic pharmacophore divide psychedelics into two broad families, tryptamines on the left side of the figure and phenethylamines on the right side. The conversed pharmacophore is highlighted in each psychedelic structure in the figure.
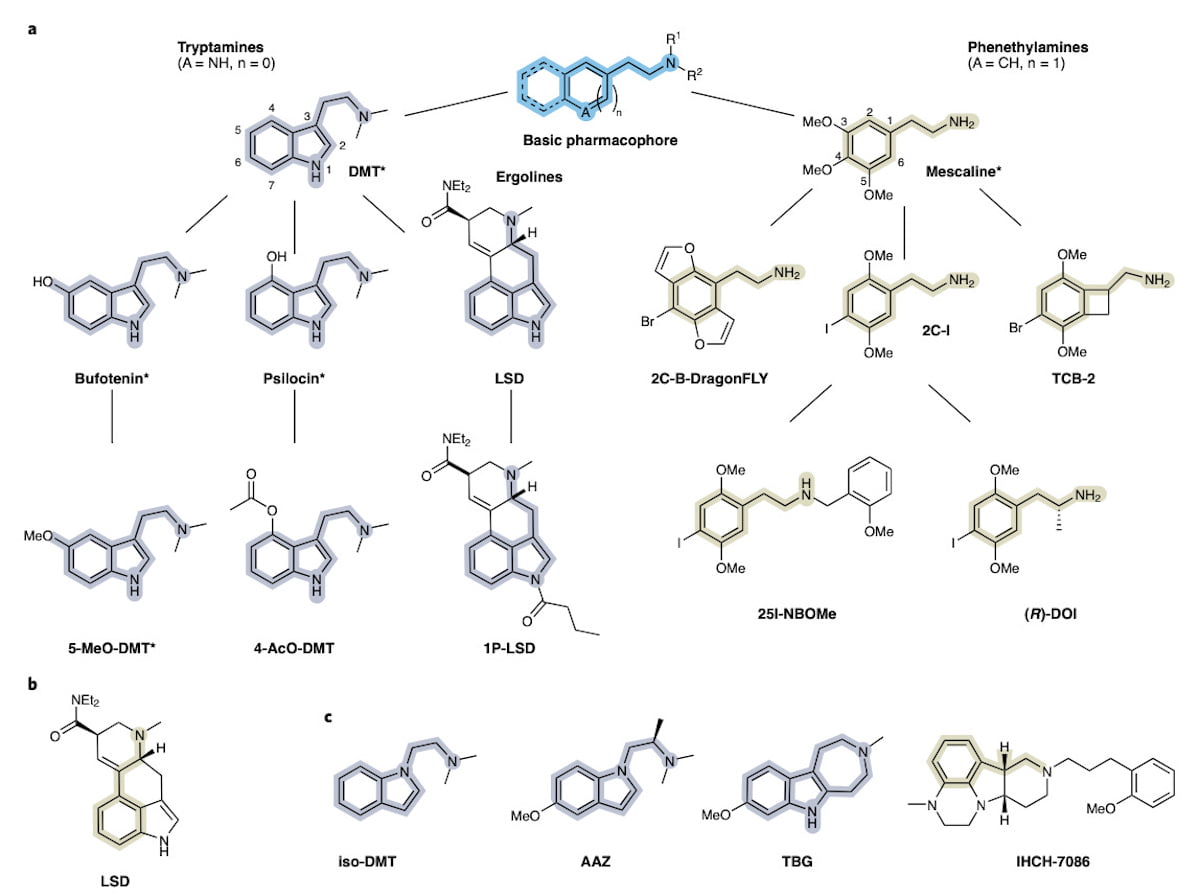
The chemical structures of psychedelics – note how the chemical structures are conserved within the two families.
Potency and other drug effects are modulated by how each psychedelic binds to the 5-HT2A receptor. Each of these different psychedelics is a different key, and they all fit into the same lock.
However, some keys fit perfectly into locks (like serotonin into its receptor). Other keys almost fit a lock, or a key might force its way into a lock and get stuck for a while. The length of time the key is in the lock and/or how tight the key/lock fit is can lead to different psychedelic experiences.
Chemical isomers
Keys can look almost identical and fit different locks. Similarly, chemical molecules called isomers can have almost the exact same chemical structure but they differ only in the position of a chemical group, which can result in different psychedelic effects.
For example, psilocin and bufotenin are both psychedelics with nearly identical chemical structures. The only thing that is different is where the alcohol (-OH) chemical group is located on the aromatic ring. The result is very different hallucinogenic effects.
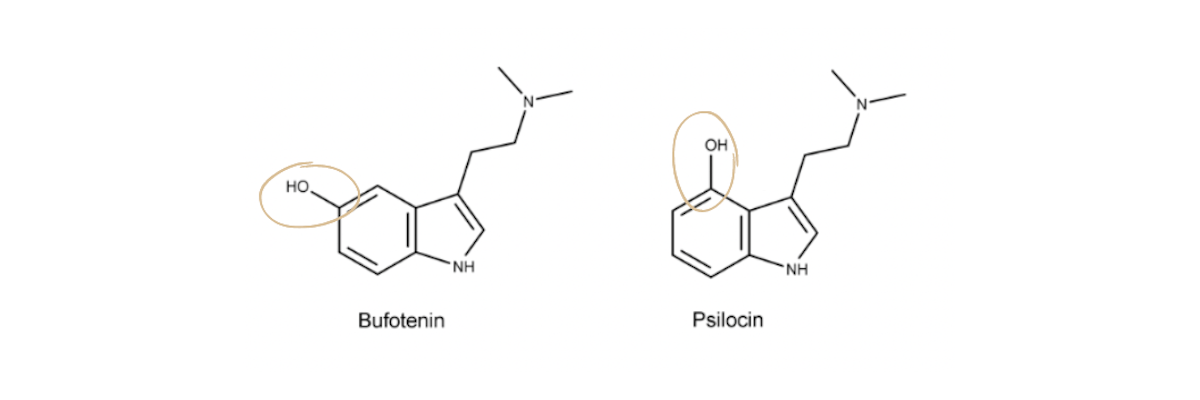
The chemical structures of Bufo and Psilocin are exactly the same, except the -OH group is in different places on the 6-pointed ring.
Psilocybin is a naturally occurring psychedelic substance found in certain types of mushrooms. When consumed, it is converted to psilocin in the body, which is thought to be responsible for its effects including altered states of consciousness, changes in perception and emotional changes. It can also cause visual and auditory hallucinations and alter the sense of time.
Bufotenin, also called Bufo or “the toad”, is a naturally occurring psychedelic substance found in the skin of some toads and in certain plants. Bufotenin is known to produce effects similar to those of other psychedelics, including altered states of consciousness, changes in perception and emotional changes. However, its effects are generally less intense and shorter-lasting than those of other psychedelics, such as psilocybin or LSD. A typical Bufo trip is about 20 minutes long.
Even these short hallucinogenic trips can be very impactful. On a Joe Rogan episode, former heavyweight boxing champion Mike Tyson described his experience with Bufo and said it profoundly changed his life.
“I look at life differently, I look at people differently. It’s almost like dying and being reborn… It’s inconceivable. I tried to explain it to some people, to my wife, I don’t have the words to explain it. It’s almost like you’re dying, you’re submissive, you’re humble, you’re vulnerable — but you’re invincible still in all.”
Stereochemistry
Furthermore, psychedelic chemicals are very complex. Just like a key, chemical molecules are 3-dimensional, and chemists call this stereochemistry. They can exist as mirror images of each other, like your right and left hands. Chemists call this chirality, and the mirror images are referred to as R- and S-enantiomers.
A mixture of the R and S enantiomers is called a racemic mixture, and the individual R or S enantiomer can be isolated to a pure substance of just one enantiomer. All three substances, the racemic, the R- and the S-enantiomer, can have different potency, psychedelic effects, or lack psychedelic effects.
You may have heard about racemic mixtures in reference to the anesthetic drug ketamine.
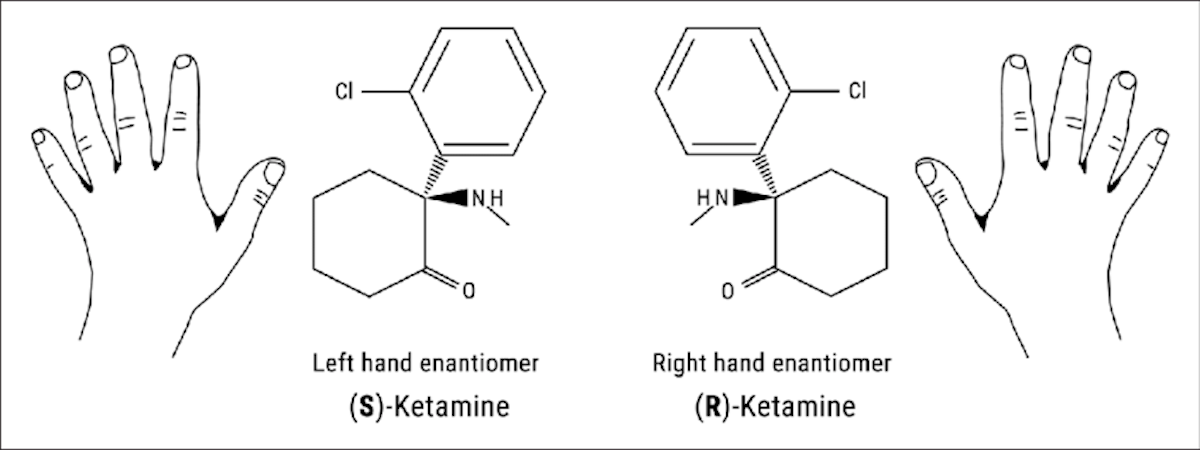
The R and S enantiomers of Ketamine. Figure from Jelen, L.A et al. “Ketamine: A tale of two enantiomers” DOI:10.1177/0269881120959644
The racemic mixture of ketamine is a mixture of the two mirror images of the molecule, and it is typically used as an anesthetic and is known to produce a range of effects, including dissociation (a feeling of disconnection from one’s body and surroundings), hallucinations, and altered states of consciousness.
“Esketamine” is the isolated S-enantiomer of ketamine, meaning scientists isolated one of the mirror-image chemicals of ketamine. Additionally, Esketamine is the pharmaceutical trade name of a nasal spray that has been approved by the US Food and Drug Administration (FDA) for the treatment of treatment-resistant depression in adults in combination with an oral antidepressant. It is thought to produce its antidepressant effects by inhibiting the action of a neurotransmitter called glutamate in a specific region of the brain called the prefrontal cortex.
Ketamine is not a classic psychedelic and acts primarily on the NMDA (N-methyl-D-Aspartate) receptor, rather than the 5-HT2A receptor.
Psychedelic effects
Now that you’re a chemistry expert, you know that tryptamines and phenethylamines have similar chemical structures to serotonin (5-HT), which explains why they bind the serotonin receptor. However, these chemicals produce different pharmacological effects than our naturally occurring serotonin.
Serotonin can influence learning, memory, happiness as well as regulating body temperature, sleep, sexual behavior and hunger. While psychedelics can produce a range of psychological effects, including altered states of consciousness, altered perception, and changes in mood and thought.
Most psychedelics bind the 5-HT2A receptor, however, that is not the only serotonin receptor. There are 14 distinct 5-HT receptors in humans, and some psychedelics, like LSD, bind most of the 14 receptors. In addition, some psychedelics will bind other neurotransmitter receptors, like the dopamine receptors. It is currently not known which of the receptors mediate the potential therapeutic actions of psychedelics, and recent studies suggest 5-HT2A is not acting alone.
Remember how the receptors act as a lock, and the psychedelics act as a key. When the psychedelic binds the receptor, it can hold the lock in an activated (unlocked) or deactivated (locked) state. The serotonin receptor acts like a dam that controls the flow of ions from outside of a brain cell (a neuron) to the inside. When the receptor is activated by a psychedelic and unlocks the dam, this leads to a flow of ions into the neuron which excites the cell and changes its usual activity.
This flow of ions in neurons in the frontal cortex of the brain, are thought to increase dendritic excitability. Dendrites are the branches of neuron cells, and psychedelics have been shown to promote the formation of new dendritic spines – called promoting neuroplasticity. Neuroplasticity is a way for the brain to rewire itself. Neuroplasticity can promote learning, development and forming new memories.
It has been demonstrated in lab-grown neuron cells that the dendritic spine size, density, and number of dendrites on each cell increased after psychedelic treatment. In another study with psilocybin, the dendritic spine density remained elevated up to 1 month after the initial administration of psilocybin.
Visual hallucinations are believed to occur in the primary visual cortex of the brain. It has been found that neuronal firing decreases in response to LSD. However little else is known.
Not only are visual hallucinations a mystery to scientists. It is also not understood how behavioral changes are linked to the biological action of psychedelics. Understanding this link would be beneficial to identifying which individuals are more likely to respond positively to psychedelic therapies.
This article was first published in Nina’s Notes on 28 December and is republished on Psychedelic Health with permission.


 Opinion2 years ago
Opinion2 years ago
 Insight3 years ago
Insight3 years ago
 Medicinal2 years ago
Medicinal2 years ago
 Medicinal2 years ago
Medicinal2 years ago
 Research2 years ago
Research2 years ago
 Markets & Industry1 year ago
Markets & Industry1 year ago
 News3 years ago
News3 years ago
 Medicinal2 years ago
Medicinal2 years ago




















