Medicinal
What we know about the effect of psychedelics on women’s health
Published
1 year agoon
By
News Editor
In this article, Nina Patrick, Ph.D. and Natasha Antropova, Ph.D., explore what the science says about psychedelics and women’s health, from hormones, depression and eating disorders to endometriosis, chronic pelvic pain, trauma and more.
Psychedelics are experiencing a renaissance. These mind-altering substances, which were once prohibited and criticized, are gaining attention again as potential remedies for various clinical problems.
The effectiveness of psychedelics has been documented by studies performed as early as the 1960s, and nowadays multiple clinical trials are underway to investigate the potential of psychedelic drugs such as ketamine, MDMA, and psilocybin mushrooms to treat various conditions, including depression, anxiety, substance abuse, post-traumatic stress disorder, eating disorders and many more. With these long-stigmatized substances making a comeback in the scientific realm, it is possible that women may see a particular benefit from them.
At present, there is a lack of understanding among scientists regarding how the pharmacology of psychedelic substances might impact a woman’s physiology. That is not surprising, since much of the past research in this area has focused on white male populations, and has left a significant gap in our understanding of how psychedelics may impact women’s health.
This presents a large opportunity for research to investigate how psychedelics may influence female-specific issues such as chronic pelvic pain, mood disorders, perinatal depression and even the challenges of motherhood.
Connection to hormones
Classic psychedelics work by activating serotonin (5-HT2A) receptors in the brain. Activation of the 5-HT2A receptors can lead to changes in mood and promote neuroplasticity, or the brain’s ability to change and adapt, which could be important to treating mental health disorders.
Serotonin is commonly associated with happiness and elevated mood, it is also involved in the modulation of pain from the brain to the spinal cord. While the mechanism is not fully understood, there is promising evidence that if you treat one of the domains of pain, anxiety, or depression, another may also improve.
The literature has also shown that female hormones, particularly estrogen, have intriguing interactions with the serotonin 5-HT2A receptor. Research has demonstrated that estrogen increases the density of 5-HT2A binding sites in areas of the brain responsible for mood, mental state, emotion, cognition and behaviour.
The relationship between estrogen levels and 5-HT2A binding sites may also provide clues to gender differences in psychiatric conditions like schizophrenia and depression, which are more common in women than men. Furthermore, disruption of estrogen levels during menopause may lead to dysregulation of the 5-HT2A and brain-derived neurotrophic factor (BDNF) signaling pathway, which could predispose the brain to depression.
Given that classical psychedelics, like psilocybin, MDMA and LSD, primarily elicit their effects via the 5-HT2A receptor, the presence, absence and combinations of female hormones could potentially enhance the effect of psychedelic compounds in women. However, the research on understanding the effects of psychedelics in the unique environment of the female body is still in its infancy.
Gender differences
Exploring gender differences in the effects of psychedelics is an important step toward making them clinically available. Various research groups in the field have looked into the effects of psilocybin on mental states and cognitive functions between male and female groups. Surprisingly, little variance was found.
A study published in June 2022 led by Zach Walsh’s research team at the University of British Columbia, Kelowna, aimed to assess the impact of psilocybin microdosing on mood and mental state. They followed 1133 individuals for two years, out of which 953 individuals engaged in their mushroom microdosing practice, and 180 individuals did not consume anything.
The individuals who microdosed mushrooms reported small to medium improvements in mood, anxiety and depression. Interestingly, no significant gender differences were found except for with depression, where women reported a higher improvement than men.
Furthermore, a separate study with rats given psilocybin found no disparities in decision-making and motivation, and no differences amongst male and female rates. Overall, these studies indicate that differences in the effects of psychedelics may not be significant between genders, but more studies are underway.
While no gender differences have been found yet, psychedelics do show promise as a clinical solution for certain disorders that are more prevalent in women. For instance, women are more susceptible to PTSD than men, and eating disorders such as anorexia nervosa are three times more common among women than men.
Let’s look at what we know so far.
Eating disorders
To date, several studies have investigated the effects of psychedelics on body dysmorphic disorder and eating disorders. The overall conclusion is that the efficacy of this treatment is unclear, but there is a lot of support and encouraging stories to warrant further exploration.
A Toronto-based group performed an extensive review of 372 studies using psychedelics for eating disorders performed before February 2022. While positive results were observed, they were mostly identified through thematic analysis and self-reports. These findings underscore the need for further exploration, which multiple groups are currently pursuing.
One group is the Multidisciplinary Association for Psychedelic Studies (MAPS), which is currently conducting a study on MDMA-assisted therapy with 36 participants suffering from anorexia, bulimia or binge-eating disorders. The Centre for Psychedelic Research at Imperial College London is also leading a clinical trial on psilocybin-assisted psychotherapy as a treatment for anorexia.
Premenstrual Dysphoric Disorder (PMDD)
Premenstrual dysphoric disorder (PMDD), a severe mood disorder, is characterized by mental health and physical symptoms in the week before the onset of menstruation and affects millions of women worldwide. It is a much more intense form of PMS.
Currently, the first line of treatment for PMDD are selective serotonin reuptake inhibitors (SSRIs). Unlike in other mood disorders, patients with PMDD experience rapid onset of action from SSRI (as opposed to the typical two to three week onset time). Other treatments include hormone therapy, psychotherapy and supplements such as calcium, vitamin B6 and magnesium.
Unfortunately, many women with PMDD do not respond to the traditional treatment options. Recent research shows that ketamine administered at the onset of PMDD may alleviate the treatment-resistance symptoms.
Ketamine is FDA-approved for use at high doses as an anaesthetic to put you asleep during surgery. Although not FDA-approved, lower doses that are “sub-anesthetic” are used “off-label” to treat depression, pain and other mental health and substance use disorders.
Recent data shows that ketamine actually activates estrogen receptors. This is important because declining levels of estrogen during the luteal phase may be a contribution factor to PMDD. By administering ketamine at the start of the luteal phase, or when symptoms of PMDD become apparent, it may be possible to treat one of the big contributing factors.
The same study showed that ketamine works with estrogen to increase glutamate levels. Glutamate is a neurotransmitter that may be uniquely connected to major depression. The lack of glutamate in certain regions of the brain has been strongly linked to depression.
Ketamine has been shown to activate glutamate transmission in the brain and is thought to be a key mechanism in its anti-depressant effects. Therefore ketamine may have a direct impact on mood and other signs of depression is administered at the onset of PMDD.
Endometriosis, chronic pelvic pain and trauma
Chronic pelvic pain affects 26% of the female population and is associated with negative cognitive, behavioral, sexual and emotional consequences which affect many aspects of a person’s life. Endometriosis is a component of 70 to 80% of the cases of chronic pelvic pain, and over 170 million women worldwide suffer from endometriosis.
Chronic pelvic pain is more severe in women with a history of physical or psychological abuse and mental illness. A study of over 60,000 pre-menopausal women found that women who reported severe sexual and physical abuse in childhood and adolescence had a 79% increased risk of endometriosis. The study suggested that early traumatic experiences affect the production of stress hormones and inflammatory responses that contribute to chronic pelvic pain and other pain symptoms.
Since serotonin is involved in the modulation of pain, anxiety, and depression, psychedelics which target the 5-HT2A receptor offer a hopeful prospect for women suffering from chronic pelvic pain, endometriosis and trauma.
At the moment, no clinical trials using psychedelics for endometriosis and chronic pelvic pain are currently listed on clinicaltrials.gov, which indicates an opportunity for research and development.
Psychedelics during pregnancy
Currently, there is a lack of clinical research on the effects of psychedelics during pregnancy. While some initial studies from the late 1960s and 1970s indicate that LSD use during pregnancy did not cause harm or congenital disability, these findings are not definitive and necessitate further investigation.
It is important to remember that the use of psychedelics during pregnancy is illegal due to the unknown impact on fetal development. Furthermore, certain psychedelics, such as LSD and mescaline, have shown the ability to provoke uterine contractions in animal studies.
Prenatal and postpartum depression
Depression is a common mental disorder among women before, during and after giving birth. Depression affects 6 to 12% of mothers in high-income countries and a larger population, 20 to 25%, of mothers in middle-income and low-income countries.
Studies have linked Cesarean sections (C-sections) to postpartum depression (PPD), which is related to reduced quality of parent-child interaction and adverse effects on maternal and child health. Cesarean section deliveries account for nearly 30% of births annually, and emergency C-sections account for 7 to 9% of all births.
Evidence is generally lacking for the use of psychedelics for pregnancy-related depression, however, due to the known safety profile of ketamine and its FDA-approval, it has been investigated for its use in prenatal and postpartum depression.
Prenatal Depression
In a collaborative effort between Peking University and the Outcomes Research Consortium in the USA, a small study was conducted to test the feasibility of ketamine use in prenatal depressive symptoms for the possibility of running a future large randomized trial.
A total of 66 women with prenatal depression who were scheduled for an elected cesarean delivery were randomized to receive either low-dose ketamine (0.5mg/kg) or placebo. The study drugs were administered intravenously over a 40-minute period after clamping the umbilical cord. Depression was assessed 48 hours postpartum.
The depression score did not differ between the groups, but the pain intensity was less severe at four hours postpartum in the ketamine group. Despite limited differences between the ketamine and placebo group on depression score, the study protocol demonstrated that it is feasible for a large randomized trial.
Postpartum depression
Ketamine was investigated for its effect on postpartum depression in women undergoing cesarean delivery. In a larger study of 654 women undergoing C-section, 10 minutes after childbirth, patients in the ketamine group were given a higher dose of ketamine, 0.5mg/kg, while the women in the control group were given standard postpartum care.
The prevalence of postpartum blues and postpartum depression were significantly lower in the ketamine group, and the study findings suggested that ketamine functions are a preventative agent against postpartum depression.
Psychedelics in motherhood
From the moment a child is born, a mother’s responsibilities grow exponentially. Her days become filled with the tasks of two people, mother and child, that require attention, care and patience. Despite the joy that motherhood brings, it can also be a stressful and overwhelming experience, exacerbated by the societal pressure on women to balance work and life, and essential “do it all.” Motherhood can leave mothers feeling drained and burnt out.
Microdosing psychedelics has emerged in recent years as a potential solution to alleviate some of the stress and anxiety of motherhood. While this approach is controversial, many mothers have reported positive experiences and benefits from microdosing. They claim that microdosing helped them manage their emotions, improve their mood and enhance their well-being.
A Colorado-based, nationwide group called Moms on Mushrooms (M.O.M), was created to teach interested moms about using a small dose of psilocybin mushrooms. The community has connected mothers who share and mirror each other’s struggles as a parent and has helped women feel comfortable enough to try microdosing.
Tracy Tee, the founder of M.O.M, told Rocky Mountain PBS: “I just really felt a calm and a space between my world that I hadn’t felt before. In the same way that you take an antidepressant, that’s what microdosing is.”
A majority of people using SSRIs, or prescription antidepressants, are women, and the World Health Organization has found major depressive disorder the leading cause of disease burden for women internationally.
Some mothers have reported that microdosing mushrooms has assisted them enough to slowly stop taking antidepressants or anti-anxiety medicines.
They say that taking a microdose, between 0.1 milligrams and 0.3 milligrams, taken over a period of days, gives them more patience, the ability to cope, more organizational talent and more happiness in the physically and emotionally difficult hours of raising children.
Many mothers also tout that a microdose of mushrooms is safer and less high-inducing than a glass of wine or beer. Many report that the microdoses are so small that they do not, or barely, register a “buzz.”
Despite these anecdotal reports of improvements, psilocybin and mushrooms remain illegal in a majority of countries and regions, but Colorado is progressive on this regulation. On November 8, 2022, Colorado passed the Natural Medicines Health Act which allows people 21 and older to grow and share psychedelic mushrooms.
Policy and progress
Psychedelic Medicines were predicted to be a gamechanger in 2023 by The Economist’s video The World Ahead 2023: five stories to watch out for. Policy reform around psychedelic therapies with the Natural Medicines Health Act in Colorado and Australia’s recent reclassification of psilocybin and MDMA to enable prescription are leading the way and setting the example for other countries to follow.
It is anticipated that MDMA-assisted therapy for the treatment of PTSD will pass FDA approval in late 2023 or early 2024, and in the next few years psilocybin is expected to be considered for FDA approval as well.
In November 2022, results from a Phase 2 psilocybin trial found that a single 25 milligram dose eased symptoms of depression for patients with treatment-resistant depression.
Although a few clinical trials have shown significant improvement for patients with certain mental health disorders, many early clinical trials, especially trials for women’s health issues are missing conclusive data on the benefits of psychedelics. There presents an opportunity for more research and development to meet these unmet needs and see if psychedelics can make an impact in women’s health.
Nina Patrick, Ph.D., is a biotech entrepreneur and content creator with expertise in psychedelics, drug discovery, diagnostics and longevity. You can reach her at Nina@ninapatrick.xyz.
Newsletter: www.notes.ninapatrick.xyz
Website: www.ninapatrick.xyz
Email: nina@ninapatrick.xyz
Twitter: @ninapatrick
Natasha Antropova, Ph.D., is an AI product manager at DeepMind and content creator with expertise in women’s health and AI. Please reach out at iam.natasha.antropova@gmail.com
newsletter: https://healthyhertech.substack.com
email: iam.natasha.antropova@gmail.com
twitter: @Natashantropova
LinkedIn: https://www.linkedin.com/in/natashaantropova
You may like
-


UK advisory body issues rapid response on psychedelics for anxiety
-


SRI and non-SRI patients experience similar antidepressant effects of psychedelics
-


Europe to establish regulatory guidance on psychedelics in 2024
-


Beckley Psytech receives $50 million investment from atai Life Sciences
-
Doctors warn against potentially harmful psychedelic “trip killers”
-


Vital report explores employment in the psychedelics industry
Medicinal
MDMA for PTSD receives priority review for New Drug Application
Published
2 months agoon
14th February 2024
Lykos Therapeutics, formerly MAPS Public Benefit Corporation, has announced it has received FDA acceptance and priority review for a New Drug Application (NDA) concerning its MDMA-assisted therapy for PTSD.
The FDA has accepted Lykos’s NDA for MDMA capsules used in combination with psychological intervention. This intervention includes psychotherapy and other supportive services provided by a qualified healthcare provider for individuals with PTSD.
Lykos has stated that the FDA has granted the application priority review and has assigned a Prescription Drug User Fee Act (PDUFA) target action date of August 11, 2024. Current statistics show that 89% of applications that meet their PDUFA deadline are approved. If this application is approved, it would make this the first MDMA-assisted therapy and psychedelic-assisted therapy.
In a press statement, Amy Emerson, chief executive officer of Lykos Therapeutics, commented: “Securing priority review for our investigational MDMA-assisted therapy is a significant accomplishment and underscores the urgent unmet need for new innovation in the treatment of PTSD.
“We remain focused on working with the FDA through the review process and preparing for a controlled launch with an emphasis on quality should this potential treatment be approved.”
The NDA submission was supported by results from several studies on the therapy, including two Phase 3 studies that looked at the efficacy and safety of the therapy. Both of these studies met their primary endpoints, which were a change in PTSD symptom severity and an improvement in functional impairment associated with PTSD.
While no serious adverse events were reported in the MDMA group in either study, Lykos highlights that the safety and efficacy of MDMA-assisted therapy have not been established for the treatment of PTSD.
The news has been welcomed across the pond by European campaign groups advocating for access to psychedelic-assisted therapy.
In a press statement, campaign group PAREA commented: “Innovation in mental health has stagnated for decades. In the past three years, Europe has approved only one new psychiatric treatment, compared to 68 in oncology.
“While the U.S. is on the brink of approving the first psychedelic-assisted therapy, Europe significantly lags behind. This is primarily because the current incentives and rewards for companies to conduct large-scale pivotal trials on psychedelics are insufficient in Europe, highlighting the need for enhanced support and incentives to advance novel mental health treatments.”
While the US makes strides in advancing psychedelic healthcare, Europe is now beginning to take note of this scientific development, with the European Medicines Agency (EMA) set to hold a multi-stakeholder workshop on medical psychedelics in April 2024.
The workshop aims to establish regulatory guidelines for the development and therapeutic use of psychedelic substances in Europe.
The continent also made a recent historic advancement in the field of psychedelic research. In January 2024, the European Union announced €6.5 million in funding for research into psychedelic therapy as part of its Horizon Europe programme.
The funding has been awarded to a consortium of 19 partners from nine different European countries for a clinical trial – the PsyPal trial – which will study psilocybin-assisted psychotherapy for psychological and existential distress in people who are diagnosed with either chronic obstructive pulmonary disorder (COPD), multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS) or atypical Parkinson’s disease (APD).
Medicinal
Doctors warn against potentially harmful psychedelic “trip killers”
Published
4 months agoon
3rd January 2024By
News Editor
Doctors have raised a warning against so-called “trip killers” that are used to end challenging psychedelic experiences on compounds such as LSD or psilocybin.
The doctors have published the warning in a letter in the Emergency Medicine Journal. In the letter, an analysis of relevant Reddit threads is provided that show drugs such as benzodiazepines and antipsychotics recommended to help end these challenging psychedelic experiences. However, the doctors emphasise that these recommendations rarely include information about potential side effects.
A total of 128 Reddit threads created were discovered that were created between 2015 and 2023, yielding a total of 709 posts. With 440 recommendations, amounting to nearly half – 46% – of all the ‘trip-killers’ mentioned in posts, were various benzodiazepines, followed by several different antipsychotics at 171%.
The team found that one in 10 recommendations were for antidepressants, while one in 20 were for alcohol. Opioids, antihistamines, herbal remedies, such as camomile and valerian, and prescribed sleeping pills, attracted 3% each, with cannabis and cannabidiol at 2%.
Trip-killers were mostly discussed in reference to countering the effects of LSD (235 recommendations), magic mushrooms (143), and MDMA (21). Only 58 posts mentioned potentially harmful side effects.
The authors write: “The popularity of benzodiazepines raises concerns. Benzodiazepines are addictive and have been repeatedly implicated in overdose deaths.
“The doses described on Reddit risk over-sedation, hypotension [low blood pressure], and respiratory depression [stopping breathing or shallow breathing].”
Doses of one of the recommended antipsychotics, quetiapine, were also high the authors note, with only a few posts differentiating between fast and slower release formulations.
“Information on trip-killers isn’t available through drug advice services, despite the probable risks they pose,” highlight the authors.
Medicinal
Integrating metaphysics into psychedelic therapy
Published
7 months agoon
29th September 2023
Dr Peter Sjöstedt-Hughes, Lecturer at Exeter University, has proposed incorporating metaphysical philosophy into psychedelic therapy to help improve therapeutic outcomes.
Sjöstedt-Hughes suggests that psychedelic therapy may gain more advantage by extending its scope into metaphysics, helping patients better integrate and understand psychedelic-induced metaphysical experiences.
Such improved outcomes may be seen if patients undergoing this therapy “are provided with an optional, additional, and intelligible schema and discussion of metaphysical options at the integrative phase of the therapy.”
In the paper, Sjöstedt-Hughes puts forward this schema as the “Metaphysics Matrix” and an accompanying “Metaphysics Matrix Questionnaire (MMQ)” which can be utilised by therapists and researchers as a tool for the quantitative measurement of a psychedelic experience.
The paper ‘On the need for metaphysics in psychedelic therapy and research’ has been published in Frontiers in Psychology.
What is metaphysics?
While mysticism deals with understanding the universe through direct experience, such as revelation, metaphysics is a branch of philosophy that deals with understanding the fundamental nature of reality through logic/argument.
Sjöstedt-Hughes writes that “metaphysics is not mysticism” but there is overlap: “[…] metaphysics is broader and its positions can be logically deliberated — as such metaphysics can encompass mystical experiences induced by psychedelic intake yet metaphysics can also ground those experiences in a manner that can be more intelligible, comprehensive, viable, and acceptable to participants than that which the framework of mysticism alone can offer.”
The Metaphysics Matrix
A number of clinical trials investigating psychedelic-assisted psychotherapy for the treatment of mental health conditions, such as anxiety and depression, report that participants who undergo a “mystical experience” during a psychedelic session often have higher levels of sustained therapeutic outcomes.
In clinical trials, mystical experiences are measured by different scales including the Mystical Experience Questionnaire (MEQ), the Hood Mysticism Scale (HMS), the Hallucinogen Rating Scale (HRS), the Five Dimensions Altered State of Consciousness Questionnaires (5D-ASC) and Eleven Dimensions Altered State of Consciousness Questionnaires (11D-ASC).
Sjöstedt-Hughes writes: “Data derived in this manner is obviously limited and abstract not only because psychedelic experience need not be “mystical,” but also because the definition of “mystical” could be expanded to include other criteria […]
“With regard to psychedelic-assisted psychotherapy […] speaking about mystical experience per se will not be sufficient to provide a meaningful explanation of the significance of such experience to a person, for the simple reason that mystical experience is the phenomenon to be explained — mystical experience is the explanandum rather than the explanation.
“It is metaphysics that is the means of explanation, the explanans of the mystical explanandum.”
The Metaphysics Matrix has been designed to provide a “menu” of metaphysical options that may help people to “frame, make sense of, and give significance to, their experiences”, and would be another tool in the belt of therapists to better understand these experiences.
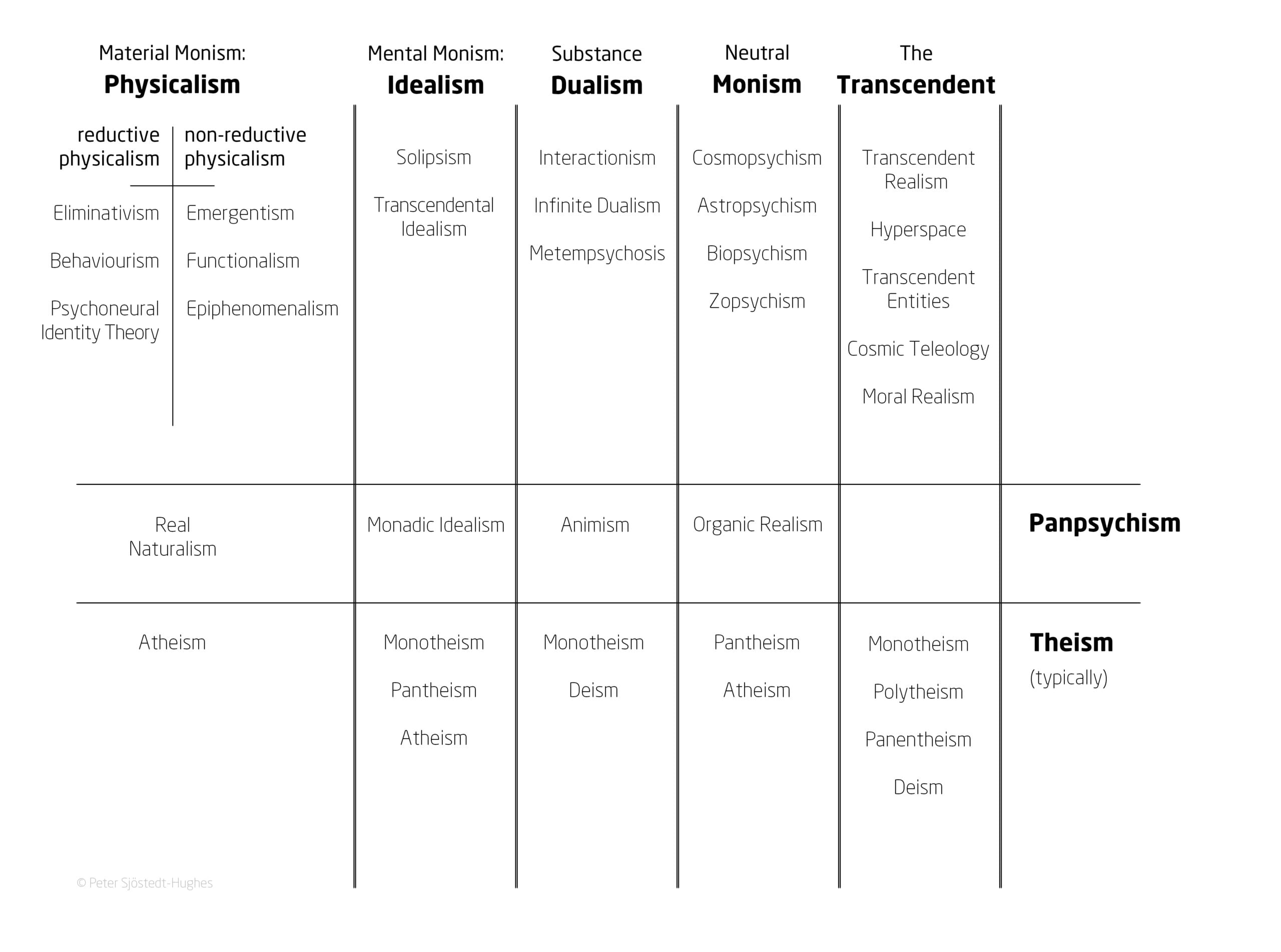
Image provided by Dr Peter Sjöstedt-Hughes.
Such experiences could be understood through metaphysical systems such as Neutral Monism, Pantheism, Panpsychism, Animism, Substance Dualism, and Idealism, says Sjöstedt-Hughes.
Some examples provided include the common experience of the Universe being God – which can be understood in the context of Pantheism – or of all matter having a basic form of sentience – such as plants having a basic drive or process – which can be understood in the context of Panpsychism.

Image provided by Dr Peter Sjöstedt-Hughes.
Additionally, enabling people who have had these experiences to understand them within these frameworks may make them less likely to dismiss the experiences as delusional, says Sjöstedt-Hughes.
“ […] Relatedly, that the worldview hitherto adopted by the participant is but one metaphysical position amongst others,” he writes.
Sjöstedt-Hughes commented: “This is a conjecture that hasn’t been tested but can be tested – offering a patient an additional and optional discussion in the integrative phase of psychedelic-assisted psychotherapy.
“Giving them this Metaphysics Menu for integration may extend the long-term benefits of psychedelic therapy and beyond because there’s a number of studies that seem to show that certain peak psychedelic experiences have the longest and most beneficial health outputs results.
“If in the integrative phase [of therapy] one looks at that experience and starts to frame it intelligibly, then the conjecture is that the participant will not in a few weeks after that, think it must have been a delusion – they will say that we don’t know what reality is.
“Therefore, we can’t dismiss something as a delusion necessarily. By doing that it might extend the significance of that experience for the person.
“When we use Mysticism Scales, by definition, mystery can’t explain itself. Metaphysics, however, incorporates those experiences and offers an explanation to what they mean. For example, the relation between oneself and the universe.”
Sjöstedt-Hughes points out that in practice, one of the immediate issues is the practical issue of implementation of Metaphysics Integration, suggesting this could be supported through resources such as a handbook or practitioner training.
He further concludes the integration would need to be “further bridged by the therapist to the participant’s life, concerns, values, aims, and outlook.”
The Metaphysics Schema is already being utilised in studies taking place at Ohio State University, US, and Exeter University, UK.
Recent Articles
- Phase 2a trial to investigate 5-MeO-DMT candidate for alcohol use disorder
- The Entourage Effect in Mushrooms: Natural psilocybin may outperform synthetic
- Psychedelics in 2024: a year for investment
- Ayahuasca retreats associated with increases in nature-relatedness
- Supporting psychedelic therapy with digital interventions
- LSD vs THC: Investigating neural complexity for altered states
Trending
Trending
-

 Opinion2 years ago
Opinion2 years agoClerkenwell Health is launching a free UK psychedelic therapist training programme
-

 Insight3 years ago
Insight3 years agoMixing psychedelics with lithium poses significant risk of seizures
-

 Medicinal2 years ago
Medicinal2 years agoMDMA therapy for PTSD granted innovation passport by UK
-

 Research2 years ago
Research2 years agoLSD trial for the treatment of adult ADHD initiated
-

 Markets & Industry1 year ago
Markets & Industry1 year agoWhere can I find training for psychedelic therapy?
-

 Medicinal2 years ago
Medicinal2 years agoMDMA: the love drug?
-

 News3 years ago
News3 years agoAwakn’s second psychedelic therapy clinic to open in London
-

 Research2 years ago
Research2 years agoLondon to host Europe’s first commercial psychedelic clinical trial facility



